 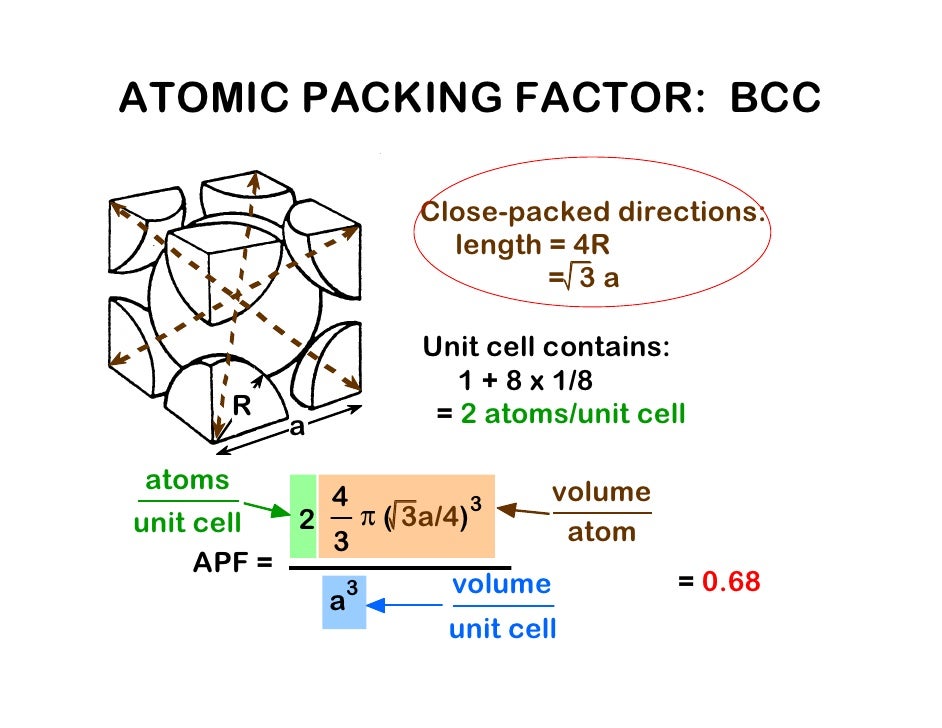
The number of particles in a bcc unit cell is 2, determined as follows: 8 spheres on the lattice corners each with an eighth oftheir volume within the cell (1 particle) and one sphere completely embedded in the lattice. To determine the edge value we must rely on the diagonal of the two opposite corners of the unit cell. So in this case the edge is greater than two atomic radii of the lattice corners. The bcc has 3 layers (along the z-axis),which would look like this: Another way of looking at the layout is using 2-dimensional layer diagrams. As the name suggests it contains an ion or atom in the center of the cube.If all the spheres have the same radius, like in metals, then the spheres centered on the lattice points do not make contact with each other. 
Rotate the body-centered cubic (bcc) unit cell. One important question should be asked: what is the efficiency of packing same size atoms in simple cubes? Because there are a total of eight one eighth volume spheres in the cell the simple cubic unit contains one net particle. But the unit cell only contains, on the lattice points, an eighth of the volume of the sphere (ion or atoms). The volume of the unit cell then is the edge cubed (edge 3). Observe that in the simple cubic cell the edge equals two atomic radii. As you rotate the spacefill model around you will notice that all the spheres (ions or atoms) are in contact with each other.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
